Using the Gibbs equation it can be said that. Its overall Ecell has to be greater than 0.

Spontaneous And Non Spontaneous Reaction Definition And Examples
A spontaneous reaction possesses a negative value for the Gibbs free energy.

. But a definitive numerical way to determine if a reaction is spontaneous or not is to look at Gibbs free energy since it represents the. Thereforeif there is a decrease in the entropy of the universe the process cannot happen. A nonspontaneous reaction will not occur unless external additions are applied to the system.
In order to tell if a reaction is spontaneous you must ask one simple question. Is there a state function of the system that can tell us whether a process. The total energy of the products of a spontaneous reaction is less than that of the reactants.
If the EMF is negative then the reaction is not spontaneous. Learn how to determine if redox reactions are spontaneous under standard conditions. The mathematical formula for Gibbs free energy is given by.
G177-T0161 Now we have to find the temperature for which this reaction is spontaneousnon-spontaneousequilibrium. Spontaneous changes also called natural processes proceed when left to themselves and in the absence of any attempt to drive them in reverse. A nonspontaneous reaction is a reaction that does not favor the formation of products at the given set of conditions.
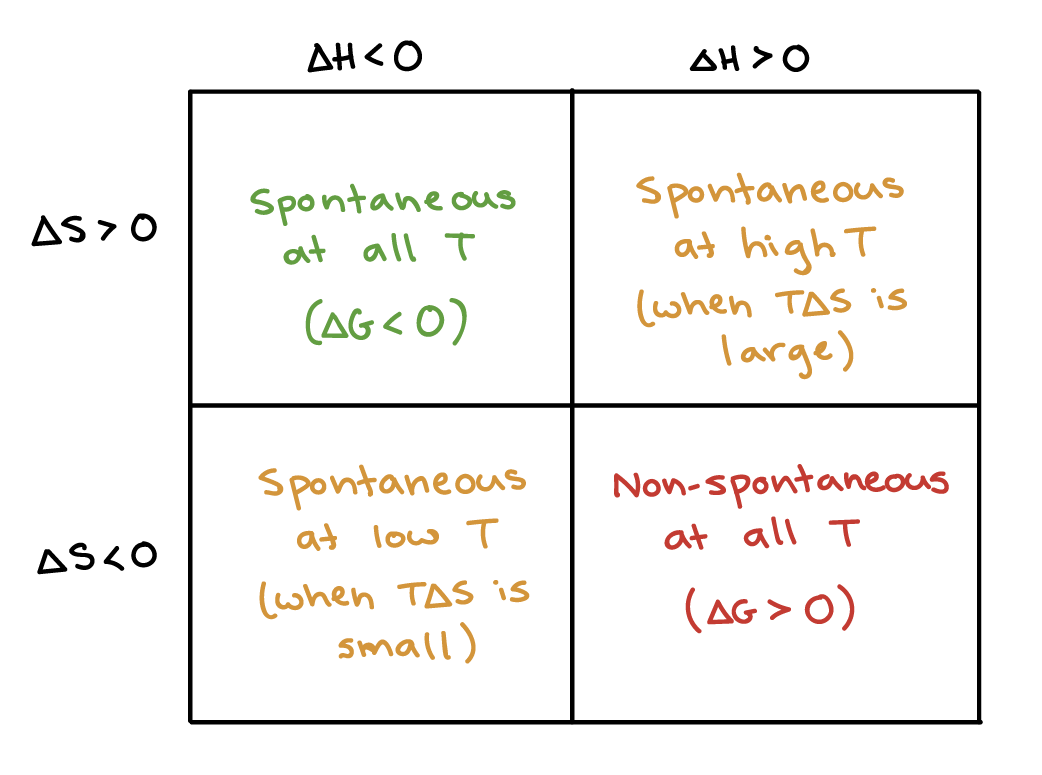
ΔS is negative entropy decreases This. To determine spontaneity of a reaction one looks at the sign of Δ G for the process. We can tell if its spontaneous if Delta G is less than 0 in other words negative we would have to see the correlation between E cell and delta G by looking at an equation that happens to be on the constants and equations sheet.
If the EMF is positive then the reaction is spontaneous. Reaction is spontaneous and goes to completion because. Reactions can be either chemical reactions or biological reactions.
There are two types of processes or reactions. The key difference between spontaneous and nonspontaneous reactions is that the spontaneous reactions have a negative Gibbs free energy whereas the non-spontaneous reactions have a positive Gibbs free energy. Total Energy of the Products.
If delta G is positive a reaction is nonspontaneous. We are allowed to have this initial spark or initial push to let the reaction go but once the reaction starts if it continues to go without any outside intervention we. If 0 the system is at equilibrium.
In order for a reaction to be nonspontaneous it must be endothermic accompanied by a decrease in entropy or both. Well combustion reactions are considered to be spontaneous however they do need a spark or something that initiates the reaction or starts the reaction. Our atmosphere is composed primarily of a mixture of nitrogen and oxygen gases.
If you need further clarification then just comment below These values can now be plugged into the Gibbs-free energy equation. To predict whether a reaction occurs spontaneously you can look at the sign of the EMF value for the cell. Review the definition of the spontaneous process explore concepts such as enthalpy entropy and Gibbs free energy and learn how to predict a spontaneous reaction.
The total energy of the products of a nonspontaneous reaction is higher than that of. If E o for the redox reaction as written is positive the reaction proceeds in the forward direction and is said to be spontaneous. Free energy delta G is a measurement of the enthalpy and entropy of a chemical reaction.
I enthalpy is minimised ΔH - ii entropy increases ΔS ii A reaction will always be nonspontaneous if. ΔGΔHTΔS Where ΔG Gibbs free energy ΔH Enthalpy change. Thus it can be inferred that any process is spontaneous if the change in Gibbs energy of the system is less than zero or else the process is not spontaneous.
We can determine if a reaction is spontaneous or non-spontaneous by looking at the Gibbs-free energy of the reaction. The entropy of the universe strives toward a maximum. If is positive the reaction is nonspontaneous it proceeds in the reverse direction.
A reactions spontaneity ie if it occurs can be deduced from the Gibbs-free energy values. One can perform experiments to predict whether a reaction will be spontaneous or not. ΔH is positive endothermic reaction AND.
The sign convention of changes in free energy follows the general convention for thermodynamic measurements. Does the entropy of the universe increase. If delta G is negative a reaction is spontaneous and will occur.
If E o for the redox reaction as written is negative the reaction DOES NOT proceed in the forward direction and is said to be non-spontaneous. If is negative the reaction is spontaneous it proceeds in the forward direction. However turning the equation around changes the sign of the standard electrode potential and can therefore turn an unfavorable reaction into one that is spontaneous or vice versa.
Therefore with the help of the above relation spontaneity of a reaction can be easily predicted. There is no net change.

What Is A Non Spontaneous Reaction Quora

Sign Of Delta G Chemistry Community

19 1 Determining Spontaneity Of A Reaction Using Ecell Values Hl Youtube
0 Comments